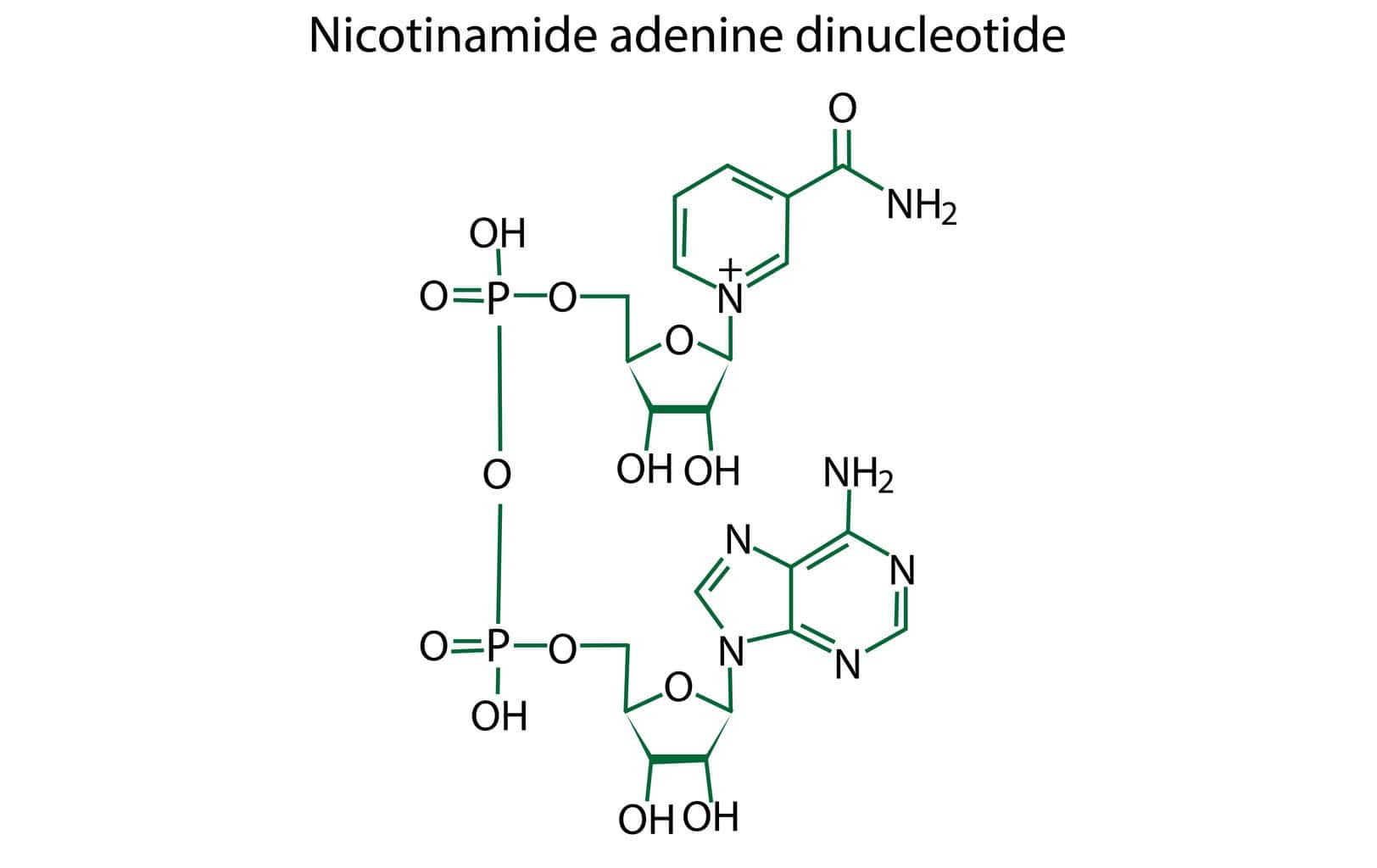
NAD+ Peptide Biosynthesis
The endogenous synthesis of NAD+ within the biological system appears to occur through a de novo pathway, which researchers posit may involve a series of enzymatic transformations of the amino acid tryptophan. This intricate process appears to comprise a sequence of several enzymatic steps. The constituents pivotal to NAD+ biosynthesis have been speculated to encompass tryptophan, nicotinamide, nicotinic acid, nicotinamide riboside, and nicotinamide mononucleotide.(1)
NAD+ Peptide Potential Functions
Upon successful biosynthesis, NAD+ has been suggested to participate in an excess of 500 enzymatic reactions and assorted cellular processes, thereby potentially facilitating and regulating a plethora of metabolic undertakings in the organism. At its core, NAD+ appears to serve as a pivotal coenzyme in redox reactions, possibly undergoing conversion into NADH and subsequently embarking on its involvement in an array of diverse metabolic pathways.(1)
Historical Background
The historical trajectory of NAD+ can be traced back to the early 1900s when Arthur Harden and William John Young made their initial discovery in 1906. They identified NAD+ as an essential cellular constituent that appears to facilitate alcohol fermentation processes.(2)
Concurrently, in the 1930s, pioneering scientist Conrad Elvehjem conducted investigations that are considered to have identified nicotinic acid and nicotinamide as precursors to vitamins. Subsequent inquiries into the biosynthesis of NAD+ in eukaryotic organisms, particularly yeast, and fungi, unveiled nicotinamide riboside as a precursor to NAD+.(3)
Mechanisms of NAD+ Action
NAD+ appears to operate as a crucial coenzyme, possibly establishing symbiotic associations with three predominant categories of enzymes: (i) the sirtuin class of deacetylase enzymes (SIRTs), (ii) poly ADP ribose polymerase (PARPs) enzymes, and (iii) cyclic ADP ribose synthase (cADPRS).(4)
SIRTs, speculated to be heavily reliant on NAD+, appears to orchestrate the regulation of mitochondrial homeostasis and stem cell rejuvenation, possibly concurrently mitigating age-associated phenomena like stem cell attrition and neuronal degradation. In parallel, the PARP consortium, which is reported to encompass 17 diverse enzymes, may catalyze the synthesis of poly ADP ribose polymers from NAD+ substrates, thereby possibly imparting genomic stability. The cyclic ADP-ribose synthases cADPRS subgroup, featuring entities such as CD38 and CD157 with possible pivotal roles in immunological processes, appears to undertake NAD+ hydrolysis, thus potentially precipitating stem cell rejuvenation and DNA repair, both considred to be vital constituents of cellular vitality.(4)
Studies suggest that, “Maintenance of NAD+ levels is important for cells with high energy demands and for proficient neuronal function. Emerging evidence suggests that NAD+ decrements occur in various tissues during aging, and that physiological and pharmacological interventions bolstering cellular NAD+ levels might retard aspects of aging and forestall some age-related diseases.”(4)
Research Studies
NAD+ Peptide and Aging
According to Nuo Sun from the Heart, Lung, and Blood Institute of the National Institutes of Health, it is imperative to regard mitochondria not solely as bioenergetics factories but rather as pivotal platforms for intracellular signaling, regulators of innate immunity, and modulators of stem cell dynamics. Sun postulates, “mitochondria are intricately intertwined with a myriad of aging-associated processes encompassing senescence, inflammation, as well as the broader age-related decline in tissue and organ performance.” In essence, mitochondria appear to emerge as the linchpin orchestrating cellular aging, and their functionality possibly constitutes a cardinal preliminary stride in comprehending how to potentially decelerate, arrest, or potentially reverse the aging course.(5)
Novel investigations propose that a subset of the age-linked degeneration observed within mitochondria might be susceptible to restoration through dietary augmentation of NAD+. This facet of NAD+ function is believed to have been initially brought to prominence, if not discovered, by David Sinclair from Harvard University, the same researcher renowned for unveiling the anti-aging attributes of resveratrol—a component present in red wine. In 2013, Sinclair illuminated that the influence of a precursor to NAD+ might possibly refurbish the youthful state of mitochondria within murine muscle tissue, thereby offering a glimpse into the potential rejuvenating capacity of NAD+.(6)
The findings of a study conducted in 2013 elucidated that with diminishing concentrations of NAD+, it may induce a pseudo-hypoxic state within cellular environments. This consequential phenomenon may possibly disrupt the customary interplay of signaling between the nucleus—housing the genetic material—and the mitochondria. Through the introduction of NAD+ supplementation to aged mice, the restoration of mitochondrial functionality was reportedly achieved. Consequently, researchers posited that NAD+ might have reinstated the intercompartmental communication pathways.(7)
NAD+ Peptide and Neuroprotection
Alterations in NAD+ dynamics may exhibit possible ramifications within the central nervous system, which appears to be intricately tied to numerous neurodegenerative ailments such as Alzheimer’s and Huntington’s.
A 2019 review article expounded on the current state of research comprehension concerning NAD+ in the context of the central nervous system. NAD+ has emerged as a lauded research compound within various murine models of neurodegenerative disorders by possibly augmenting mitochondrial functionality and potentially concomitantly abating reactive oxygen species (ROS) production. These ROS species are speculated to instigate damage in diverse inflammatory and pathological contexts, which appear to exacerbate the aging trajectory further. Moreover, there is burgeoning interest in the potential synergistic combination of NAD+ supplementation in conjunction with PARP inhibitors—a class of pharmaceutical agents involved in DNA repair and apoptotic processes. While activated PARP is integral for DNA repair, excessive PARP activity may culminate in cellular energy depletion and programmed cell death.
Research findings from murine models of Parkinson’s disease indicate that NAD+ supplementation may possibly furnish protective action against motor deficits and the demise of dopaminergic neurons within the substantia nigra. This engenders the premise that NAD+ may potentially alleviate not only Parkinson’s symptoms but also potentially retards or even forestalls disease onset.
Exploration into the kynurenine pathway (KP), a metabolic process, has unveiled intriguing prospects tied to NAD+ supplementation. Researchers have suggested that NAD+ supplementation might avert neurotransmitter breakdown and diminish the diversion of protein precursors towards NAD+ synthesis. Tryptophan amino acid, speculated to be foundational to neurotransmitter and protein synthesis, appears to be catabolized via the KP to engender NAD+. Studies state that “imbalances in absolute and relative levels of KP metabolites have been strongly associated with neurodegenerative disorders including Huntington’s, Alzheimer’s, and Parkinson’s diseases. The KP has also been implicated in the pathogenesis of other brain disorders (e.g. schizophrenia, bipolar disorder), as well as several cancers and autoimmune disorders such as HIV.” The ongoing scientific inquiry endeavors to ascertain whether NAD+ supplementation might rectify KP imbalances and possibly mitigate or even preclude the neurodegenerative disorders enumerated.(8)
NAD+ Peptide and Addiction
The adverse impacts of drugs and alcohol on NAD+ concentrations have been under speculation for some time. This negative impact is generally suggested to not only to incite nutritional imbalances but may also establish associations with alterations in affective states and cognitive faculties.
Research studies exploring the possibility of rectifying these deficiencies through NAD+ supplementation were initiated in the 1960s. However, they have garnered fresh contemporary traction owing to empirical investigations highlighting the proposed synergistic potential of NAD+ in conjunction with specific amino acid complexes. Scientific inquiries appear to ascertain that the amalgamation of NAD+ and select amino acids may bear some capacity to mitigate cravings while potentially also ameliorating stress and anxiety levels. Research in this area is still ongoing.(9)
NAD+ Peptide and Inflammation
NAMPT enzymatic entity has been associated with inflammation and appears to exhibit heightened expression in certain malignancies. Its involvement possibly extends to the pathogenesis of obesity, type 2 diabetes, and non-alcoholic fatty liver disease. The supposed capacity of NAMPT to induce inflammation appears to become more pronounced as the levels of NAD+ decrease proportionally. The proposition surfaces that NAD+ supplementation may potentially curtail NAMPT activation, possibly exerting a modulatory influence on inflammation dynamics.(11)
Conclusion
NAD+ peptides represent a promising avenue of research within the context of upregulating metabolic processes. Research findings suggest that enhancing NAD+ levels, coupled with their targeted delivery mechanisms, may possibly mitigate the impact of various age-related physiological challenges. These studies suggest an intricate interplay between NAD+ and cellular processes, including energy production, DNA repair, and gene expression regulation, which underscores the potential role of NAD+ peptides in promoting cellular vitality and longevity. Continued rigorous investigation is necessary to fully comprehend the compound’s mechanisms, and determine efficacy within specific contexts.
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.
References:
- Lymphocyte. National Human Genome Research Institute. https://www.genome.gov/genetics-glossary/Lymphocyte
- Harden, A; Young, WJ (24 October 1906). “The alcoholic ferment of yeast-juice Part II.–The coferment of yeast-juice”. Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character. 78 (526): 369–375. https://royalsocietypublishing.org/doi/10.1098/rspb.1906.0070
- Bieganowski P, Brenner C. Discoveries of nicotinamide riboside as a nutrient and conserved NRK genes establish a Preiss-Handler independent route to NAD+ in fungi and humans. Cell. 2004 May 14;117(4):495-502. https://pubmed.ncbi.nlm.nih.gov/15137942/
- Fang, E. F., Lautrup, S., Hou, Y., Demarest, T. G., Croteau, D. L., Mattson, M. P., & Bohr, V. A. (2017). NAD+ in Aging: Molecular Mechanisms and Translational Implications. Trends in molecular medicine, 23(10), 899–916. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7494058/
- Sun N, Youle RJ, Finkel T. The Mitochondrial Basis of Aging. Mol Cell. 2016 Mar 3;61(5):654-666. doi: 10.1016/j.molcel.2016.01.028. PMID: 26942670; PMCID: PMC4779179. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4779179/
- Chini CCS, Tarragó MG, Chini EN. NAD and the aging process: Role in life, death and everything in between. Mol Cell Endocrinol. 2017 Nov 5;455:62-74. doi: 10.1016/j.mce.2016.11.003. Epub 2016 Nov 5. PMID: 27825999; PMCID: PMC5419884. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5419884/
- Gomes AP, Price NL, Ling AJ, Moslehi JJ, Montgomery MK, Rajman L, White JP, Teodoro JS, Wrann CD, Hubbard BP, Mercken EM, Palmeira CM, de Cabo R, Rolo AP, Turner N, Bell EL, Sinclair DA. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013 Dec 19;155(7):1624-38. doi: 10.1016/j.cell.2013.11.037. PMID: 24360282; PMCID: PMC4076149. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4076149/
- Maddison DC, Giorgini F. The kynurenine pathway and neurodegenerative disease. Semin Cell Dev Biol. 2015 Apr;40:134-41. doi: 10.1016/j.semcdb.2015.03.002. Epub 2015 Mar 12. PMID: 25773161. https://pubmed.ncbi.nlm.nih.gov/25773161/
- NAD Therapy. https://www.addictioncenter.com/treatment/nad-therapy/
- Garten A, Schuster S, Penke M, Gorski T, de Giorgis T, Kiess W. Physiological and pathophysiological roles of NAMPT and NAD metabolism. Nat Rev Endocrinol. 2015 Sep;11(9):535-46. doi: 10.1038/nrendo.2015.117. Epub 2015 Jul 28. PMID: 26215259. https://pubmed.ncbi.nlm.nih.gov/26215259/
- Garten A, Schuster S, Penke M, Gorski T, de Giorgis T, Kiess W. Physiological and pathophysiological roles of NAMPT and NAD metabolism. Nat Rev Endocrinol. 2015 Sep;11(9):535-46. doi: 10.1038/nrendo.2015.117. Epub 2015 Jul 28. PMID: 26215259. https://pubmed.ncbi.nlm.nih.gov/26215259/