GLP-1, a concise peptide hormone spanning merely 30 amino acids,(1) reportedly orchestrates multifaceted physiological functions considered crucial for metabolic homeostasis and beyond. Studies report that its cardinal role may lie in the modulation of blood sugar levels through the augmentation of insulin secretion, thereby potentially safeguarding pancreatic beta-cell insulin stores. Moreover, GLP-1 is speculated to possess remarkable efficacy in curbing appetite, possibly by orchestrating delays in gastric emptying and mitigating intestinal motility.
A synthetic GLP-1 analog, Semaglutide peptide appears to mirror the endogenous GLP-1 peptide. Research suggests that the peptide may enhance insulin production, mitigate blood sugar levels, and preserve pancreatic beta cells by instigating insulin gene transcription. Additionally, research in Semaglutide suggest the peptide may act in appetite regulation through its purported capacity to impede gastric emptying.
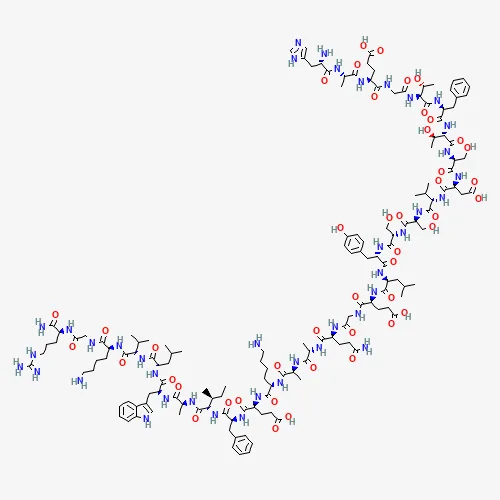
Fig. 1 – Semaglutide Peptide Chemical Structure
Research underscores the possibly profound impact of GLP-1, and by extension, Semaglutide, across a spectrum of vital organs encompassing the heart, kidneys, lungs, and liver.(2) Mechanistically, Semaglutide’s actions are postulated to unfold via intricate pathways, including its plausible interaction with GLP-1 receptors, culminating in glucose-dependent insulin release. Furthermore, Semaglutide may exert its influence by modulating glucagon release, curtailing hepatic glucose synthesis, and potentially enhancing pancreatic beta cell functionality.(3)(4)(5)
Research
Semaglutide Peptide and the Incretin Effect
Incretins, a cluster of metabolic hormones discharged by the gastrointestinal (GI) tract, are believed to instigate a reduction in blood glucose levels. As per Dr. J. J Holst, “The main actions of GLP-1 are to stimulate insulin secretion (i.e., to act as an incretin hormone) and to inhibit glucagon secretion, thereby contributing to limit postprandial glucose excursions.”
Glucagon-like peptide-1 (GLP-1) reportedly emerges as a pivotal player in this cohort, alongside glucose-dependent insulinotropic polypeptide (GIP), eliciting the incretin effect, particularly in murine models. Despite circulating at levels approximately one-tenth of GIP, GLP-1 appears to showcase superior potency, especially under heightened blood glucose concentrations.
Studies propose that the discernment of a GLP-1 receptor on the pancreatic beta cell surface may elucidate its direct role in fostering insulin exocytosis from the pancreas. Concomitant exposure with sulfonylurea compounds appears to amplify insulin secretion to an amount sufficient to induce mild hypoglycemia.(6)
This escalation in insulin output reportedly precipitates a cascade of trophic consequences, including heightened protein synthesis, attenuation of protein breakdown, and augmented amino acid uptake by skeletal muscle.
Semaglutide Peptide and Appetite Suppression
Studies conducted in murine models suggest that the introduction of glucagon-like peptide-1 (GLP-1), along with its structurally analogous counterpart GLP-1, directly into the cerebral regions of mice may result in a dampened urge to ingest food and consequent inhibition of food consumption.(7) This phenomenon suggests a potential role for GLP-1 in augmenting sensations of satiety, thereby fostering a sense of fullness and indirectly mitigating hunger.
Recent preclinical investigations have further speculated that twice-daily exposure of GLP-1 receptor agonists in murine models may elicit a gradual, linear decline in body mass. This sustained weight reduction, observed over an extended duration, is speculated to correlate with notable enhancements in cardiovascular risk profile and a concomitant decrease in hemoglobin A1C levels. The latter parameter serves as a surrogate indicator for the severity of diabetes and the efficacy of blood glucose regulation.(8)
Semaglutide Peptide and Pancreatic Beta Cells
Accumulated data from animal studies suggest the potential role of glucagon-like peptide-1 (GLP-1) in potentially promoting the expansion and proliferation of pancreatic beta cells. Moreover, there are speculations that GLP-1 may possibly instigate the differentiation of developing beta cells from progenitor cells within the pancreatic duct epithelium. Notably, research elucidates GLP-1’s reported capacity to impede beta cell apoptosis,(6) thereby tilting the delicate equilibrium between beta cell growth and demise towards proliferation. This paradigm shift suggests the peptide’s utility in diabetes management research and in safeguarding the pancreas against injuries that may jeopardize beta cell integrity.
In a noteworthy study,(9) GLP-1 appeared to exhibit some efficacy in thwarting beta cell demise induced by escalated levels of inflammatory cytokines. Intriguingly, murine models of type 1 diabetes unveil the possible protective mantle of GLP-1 over islet cells, positioning the compound as a promising avenue for further research in preempting the onset of type 1 diabetes.
Semaglutide Peptide and Neurobiological Action
Emerging data indicates a potential role for glucagon-like peptide-1 (GLP-1) in augmenting cognitive processes and shielding neurons against the ravages of neurodegenerative disorders such as Alzheimer’s disease.
Research(10) suggests that in murine models, GLP-1 appears to heighten associative and spatial learning abilities and possibly ameliorate learning impairments in mice harboring specific genetic anomalies. Moreover, scientists state that “rats overexpressing GLP-1R in the hippocampus show improved learning and memory. GLP-1R-deficient mice also have enhanced seizure severity and neuronal injury after kainate [exposure], with an intermediate phenotype in heterozygotes and phenotypic correction after Glp1r gene transfer in hippocampal somatic cells.”
Further investigations(11) in rodent models suggest GLP-1’s possible neuroprotective role against excitotoxic neuronal injury, reportedly conferring complete safeguarding of rat cohorts from glutamate-induced apoptosis—a hallmark of neurodegeneration. GLP-1 appears to exhibit a propensity for stimulating neurite outgrowth in cultured cell populations.
Both GLP-1 and its analog appear to elicit a reduction in amyloid-beta levels within murine cerebral milieu, along with potentially mitigating the presence of beta-amyloid precursor protein within neurons. Given the centrality of amyloid-beta in the formation of Alzheimer’s-associated plaques, albeit without definitive causative linkage, this observation hints at a potential avenue for mitigating Alzheimer’s disease pathology. Although the efficacy of impeding amyloid-beta accumulation in forestalling Alzheimer’s disease remains a area of conjecture, reports state that “these findings identify a novel neuroprotective/neurotrophic function of GLP-1 and suggest that such peptides may have potential for halting or reversing neurodegenerative processes in CNS disorders, such as Alzheimer’s disease, and in neuropathies associated with type 2 diabetes mellitus.”
Semaglutide Peptide and Cardioprotection
Scientific data suggests the widespread distribution of GLP-1 receptors throughout the myocardium, where they reportedly orchestrate pivotal roles in optimizing cardiac performance under specific physiological circumstances.
GLP-1’s intervention may manifest in an augmented heart rate and a concomitant reduction in left ventricular end-diastolic pressure (LVEDP). While seemingly subtle, the attenuation of LVEDP assumes significance, given its correlation with left ventricular hypertrophy, cardiac remodeling, and eventual progression to heart failure.
Recent investigations(12) suggest that GLP-1 may play a role in mitigating the harmful aftermath of myocardial infarction. The peptide is speculated to enhance the cardiac muscle glucose uptake, potentially furnishing ischemic myocardial cells with the requisite nourishment to sustain viability and circumvent apoptotic demise. This augmentation in glucose uptake is believed to operate independently of insulin, underscoring the multifaceted nature of GLP-1’s cardioprotective potential.
Experimental interventions(13) involving sizable GLP-1 infusions in canine models have yielded promising outcomes characterized by improved left ventricular performance and a concomitant reduction in systemic vascular resistance. The reported attendant reduction in blood pressure appears to not only alleviate cardiac workload but may also potentially mitigate the long-term sequelae of hypertension, including left ventricular remodeling, vascular hypertrophy, and the specter of heart failure. Dr. Holst posits that GLP-1 exposure post-cardiac injury has “constantly increased myocardial performance both in experimental animal models.”
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.
References:
- National Center for Biotechnology Information (2024). PubChem Compound Summary for CID 16135499, Glucagon-like peptide 1. https://pubchem.ncbi.nlm.nih.gov/compound/Glucagon-like-peptide-1.
- Mahapatra MK, Karuppasamy M, Sahoo BM. Semaglutide is a glucagon like peptide-1 receptor agonist with cardiovascular benefits for the management of type 2 diabetes. Rev Endocr Metab Disord. 2022 Jun;23(3):521-539. doi: 10.1007/s11154-021-09699-1. Epub 2022 Jan 7. PMID: 34993760; PMCID: PMC8736331. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8736331/
- Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol (Lausanne). 2019 Apr 12;10:155. doi: 10.3389/fendo.2019.00155. PMID: 31031702; PMCID: PMC6474072. https://pubmed.ncbi.nlm.nih.gov/31031702/
- Ahmann AJ, Capehorn M, Charpentier G, Dotta F, Henkel E, Lingvay I, Holst AG, Annett MP, Aroda VR. Efficacy and Safety of Once-Weekly Semaglutide Versus Exenatide ER in Subjects With Type 2 Diabetes (SUSTAIN 3): A 56-Week, Open-Label, Randomized Clinical Trial. Diabetes Care. 2018 Feb;41(2):258-266. doi: 10.2337/dc17-0417. Epub 2017 Dec 15. PMID: 29246950. https://pubmed.ncbi.nlm.nih.gov/29246950/
- Christou GA, Katsiki N, Blundell J, Fruhbeck G, Kiortsis DN. Semaglutide as a promising antiobesity drug. Obes Rev. 2019 Jun;20(6):805-815. doi: 10.1111/obr.12839. Epub 2019 Feb 15. PMID: 30768766. https://pubmed.ncbi.nlm.nih.gov/30768766/
- Nauck MA, Meier JJ. The incretin effect in healthy individuals and those with type 2 diabetes: physiology, pathophysiology, and response to therapeutic interventions. Lancet Diabetes Endocrinol. 2016 Jun;4(6):525-36. doi: 10.1016/S2213-8587(15)00482-9. Epub 2016 Feb 12. PMID: 26876794. https://pubmed.ncbi.nlm.nih.gov/26876794/#:~:text=The%20incretin%20effect%20describes%20the,of%20glycaemia%2C%20in%20healthy%20individuals.
- Tang-Christensen M, Larsen PJ, Thulesen J, Rømer J, Vrang N. The proglucagon-derived peptide, glucagon-like peptide-2, is a neurotransmitter involved in the regulation of food intake. Nat Med. 2000 Jul;6(7):802-7. doi: 10.1038/77535. PMID: 10888930. https://pubmed.ncbi.nlm.nih.gov/10888930/#:~:text=Pharmacological%20and%20behavioral%20studies%20confirmed,effects%20on%20body%20weight%20homeostasis.
- Blonde L, Klein EJ, Han J, Zhang B, Mac SM, Poon TH, Taylor KL, Trautmann ME, Kim DD, Kendall DM. Interim analysis of the effects of exenatide treatment on A1C, weight and cardiovascular risk factors over 82 weeks in 314 overweight patients with type 2 diabetes. Diabetes Obes Metab. 2006 Jul;8(4):436-47. doi: 10.1111/j.1463-1326.2006.00602.x. PMID: 16776751. https://pubmed.ncbi.nlm.nih.gov/16776751/#:~:text=Conclusion%3A%20In%20summary%2C%2082%20weeks,in%20some%20cardiovascular%20risk%20factors.
- Yang Z, Chen M, Carter JD, Nunemaker CS, Garmey JC, Kimble SD, Nadler JL. Combined treatment with lisofylline and exendin-4 reverses autoimmune diabetes. Biochem Biophys Res Commun. 2006 Jun 9;344(3):1017-22. doi: 10.1016/j.bbrc.2006.03.177. Epub 2006 Apr 5. PMID: 16643856. https://pubmed.ncbi.nlm.nih.gov/16643856/
- During MJ, Cao L, Zuzga DS, Francis JS, Fitzsimons HL, Jiao X, Bland RJ, Klugmann M, Banks WA, Drucker DJ, Haile CN. Glucagon-like peptide-1 receptor is involved in learning and neuroprotection. Nat Med. 2003 Sep;9(9):1173-9. doi: 10.1038/nm919. Epub 2003 Aug 17. PMID: 12925848. https://pubmed.ncbi.nlm.nih.gov/12925848/
- Perry T, Haughey NJ, Mattson MP, Egan JM, Greig NH. Protection and reversal of excitotoxic neuronal damage by glucagon-like peptide-1 and exendin-4. J Pharmacol Exp Ther. 2002 Sep;302(3):881-8. doi: 10.1124/jpet.102.037481. PMID: 12183643. https://pubmed.ncbi.nlm.nih.gov/12183643/
- Bose AK, Mocanu MM, Carr RD, Brand CL, Yellon DM. Glucagon-like peptide 1 can directly protect the heart against ischemia/reperfusion injury. Diabetes. 2005 Jan;54(1):146-51. doi: 10.2337/diabetes.54.1.146. PMID: 15616022. https://pubmed.ncbi.nlm.nih.gov/15616022/
- Nikolaidis LA, Elahi D, Hentosz T, Doverspike A, Huerbin R, Zourelias L, Stolarski C, Shen YT, Shannon RP. Recombinant glucagon-like peptide-1 increases myocardial glucose uptake and improves left ventricular performance in conscious dogs with pacing-induced dilated cardiomyopathy. Circulation. 2004 Aug 24;110(8):955-61. doi: 10.1161/01.CIR.0000139339.85840.DD. Epub 2004 Aug 16. PMID: 15313949. https://pubmed.ncbi.nlm.nih.gov/15313949/
- Image source: National Center for Biotechnology Information (2024). PubChem Compound Summary for CID 16135499, Glucagon-like peptide 1. https://pubchem.ncbi.nlm.nih.gov/compound/Glucagon-like-peptide-1.