ARA-290 was developed by researchers to specifically bind to the EPO receptor (EPOR) on non-erythroid cells, such as neurons, endothelial cells, and immune cells. Unlike EPO, which scientists consider to primarily stimulate erythropoiesis, ARA-290 does not appear to promote the production of red blood cells.
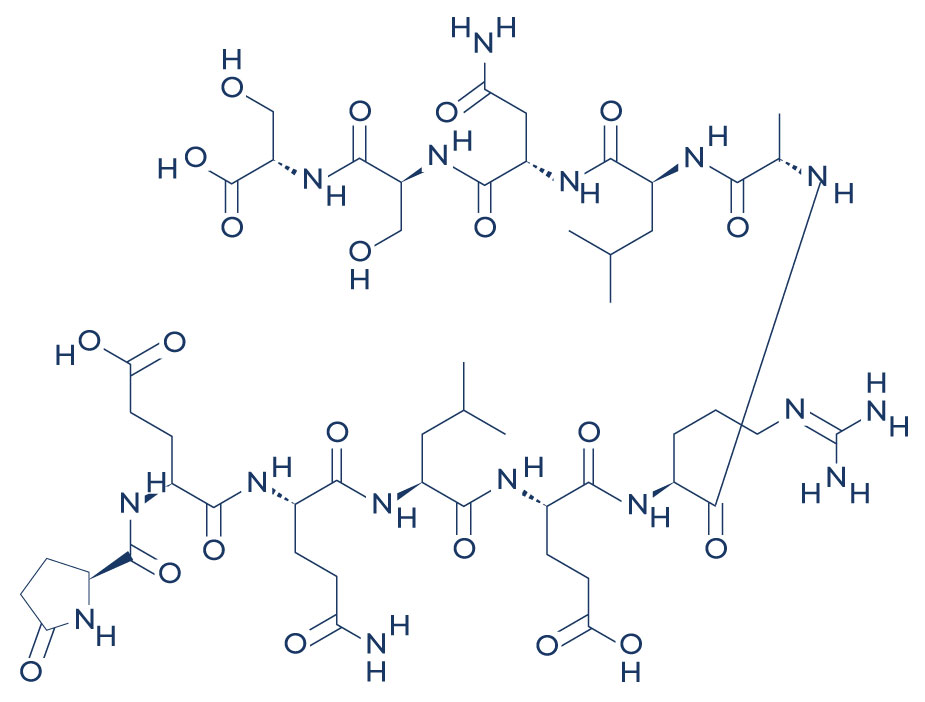
Researchers studying the peptide suggest that the ARA-290 peptide binds specifically to the IRR and may activate downstream signaling events. By activating the IRR pathway, ARA-290 potentially promotes tissue repair and reduces inflammation, which may help attenuate pain caused by tissue injury. Several preclinical and clinical studies have suggested that ARA-290 may effectively reduce pain associated with nerve injury, allodynia, and inflammation.(3)
Researchers Michael Brines and Anthony Cerami state in their study, “On administration, these agents successfully substitute for a relative deficiency of EPO production in damaged tissues in multiple animal models of disease and may pave the way to effective treatment of a wide variety of insults that cause tissue injury, leading to profoundly expanded lesions and attendant, irreversible sequelae.”(3)
Research Studies on ARA-290 Peptide
ARA-290 and Diabetic Wound Repair
Diabetic foot ulcers (DFUs) are a serious problem in diabetes and are considered to lead to delayed healing, sepsis, and ultimately amputation. The innate repair receptor (IRR) appears to be activated in injured tissue by erythropoietin, and action along this pathway may benefit diabetic wound healing.
A study evaluated the potential of ARA-290, considered a specific IRR agonist, on skin wound repair in diabetic rats. The introduction of ARA-290 to subjects once daily for 14 days appeared to result in accelerated wound closure, decreased epithelialization time, increased collagen and protein content, improved biochemical parameters, increased antioxidant levels, decreased inflammatory cytokine levels, and reduced lipid peroxidation in the rat models. The results suggest that ARA-290-mediated IRR activation may signal downstream action in wound repair. The 20% ARA-290 concentration appeared to induce the most significant action on wound contraction, collagen, and protein content compared to other groups.(4)
Mashreghi M. et al. stated, “The data confirmed wound healing activities via macroscopic, biochemical, immunofluorescent and molecular methods. There was a meaningful acceleration in wound closure rate, a decrease in the period of re-epitalization, and a greater amount of collagen and protein content in the ARA-290 peptide-treated group compared to the control group. The increase of serum insulin and HDL was divergent with blood glucose decrease and reduced lipid level. Reduced levels of inflammatory cytokines and lipid peroxidation and augmented antioxidants confirmed the healing effect. The results propose that ARA-290-arbitrated IRR activation may signify an appealing approach to treat diabetes-associated wound healing.” (4)
ARA-290 Peptide and Cardiovascular System
Multiple studies have explored the potential of ARA-290 peptide in various possible modes, including improving systolic cardiac function, reducing cardiac tissue inflammation, enhancing resistance to oxidative stress, preserving protein quality control, and augmenting mitochondrial function in cardiac myocytes.
It has been suggested by the researchers in these studies that ARA-290 peptide may attenuate the age-associated decline in cardiac function, reduce systemic inflammation, and preserve late-life body weight, ultimately reducing frailty and improvement in the health span in rats of advanced age.(5)
ARA-290 and Retinal Ischemia
A recent study has suggested that ARA-290 peptide may exhibit the potential to protect endothelial blood vessels and combat retinal ischemia, which is a leading cause of blindness. Retinal ischemia is often considered an endpoint of several diseases threatening vision. One strategy to overcome this condition may be to restore endothelial colony-forming (ECF) cells in retinal cells.
In this study, experimental mouse models of retinal ischemia underwent ECF cell transplantation to evaluate the action of the peptide. The transplantation occurred in the presence or absence of ARA-290 to determine the peptide’s potential. The study found that the peptide may have reduced the inflammatory expression of interleukin cells in the retina. Following the transplantation, the peptide appeared to yield minimal inflammation and faster recovery.(6)
Immunomodulatory Potential of ARA-290 Peptide
Alteration of Adaptive Immunity
Adaptive immunity is considered to develop over time due to increased exposure to antigens. ARA-290 peptide may alter the presentation of antigens by dendritic cells, according to research theories, thereby altering adaptive immunity. While this may appear to be a “negative” action, it may be useful in transplantation. The potential of the peptide to “fine-tune” antigens may prevent immune cells from rejecting transplanted tissues.(7)
Autoimmune Considerations
Various studies have explored the possible action of ARA-290 on various autoimmune disorders. A study(8) was conducted on mice with colitis that were given a placebo or ARA-290 peptide. While the placebo mice showed no impact, the mice presented with ARA-290 peptide exhibited apparent outcomes, including enhanced tissue quality, optimal weight gain, and better prognosis. Researchers suggested that the peptide bound with the IRR receptor cells and yielded anti-inflammatory action, possibly reducing disease progression in the mice.
In addition to research on colitis, studies have investigated the peptide’s potential for other issues, including SLE. SLE is an autoimmune disorder that is considered to cause skin rashes, joint problems, and sometimes kidney failure. Studies suggest that the ARA-290 peptide may possibly suppress autoantibodies, primarily the ANA antibodies. These antibodies appear to serve as SLE markers, and their levels may indicate the severity of the disorder, with higher levels indicating more severity. The results of this study indicated that the peptide may mitigate some kidney damage, potentially improving SLE-associated fatality rates downstream.
ARA-290 Peptide and Sarcoidosis
Scientists characterize Sarcoidosis as an abnormal accumulation of inflammatory cells, causing lumps in the skin and lungs, leading to rashes and cough. Associated with Sarcoidosis is also small nerve fiber loss and damage (SNFLD), which may lead to the dysfunction of vital bodily organs.
An initial clinical trial(1) was conducted on test subjects with Sarcoidosis where the impact of ARA-290 peptide was monitored after three consecutive presentations were delivered to subjects in one week. After the study, a 50% improvement was observed in all subjects. A follow-up trial was conducted in test subjects with Sarcoidosis with mild SNFLD symptoms. These subjects underwent ARA-290 peptide presentation 3 times a week for four weeks. After the study, researchers reported that the subjects showed significant improvement.
The most recent study involved 38 subjects (with a mean age of 50) with Sarcoidosis and associated SNFLD for almost 8 years. The researchers sought to explore the impact of ARA-290 peptide, if any, on the subjects’ symptoms and condition. The study lasted for 16 weeks, including analysis and follow-up. Once the study was completed, it was suggested by the research team that the peptide exhibited a positive prognosis, with improvement reported in all subjects.
ARA-290and Glucose and Lipid Metabolism
A recent study(4) evaluated the potential of ARA-290 on glucose and lipid metabolism in diabetic rats. The diabetic control group exhibited a reported increase in blood glucose levels from day 1 compared to the normal control group. However, introductions of ARA-290 peptide with 10% and 20% concentrations appeared to have possibly reduced glucose levels in diabetic rats. Furthermore, after 14 days, insulin levels in the serum of diabetic control rats had reportedly reduced compared to normal control rats.
The study’s results stated that the 10% and 20% ARA-290 peptide concentrations may have resulted in an apparent increase in insulin levels in diabetic rats compared to the diabetic control group. In addition, the diabetic control group reportedly exhibited increased serum lipid levels (TC, TG, and LDL) and a notable decrease in HDL levels compared to the normal control group. Conversely, 10% and 20% ARA-290 peptide presentation in diabetic rats may have resulted in a significant decrease in LDL, TC, and TG levels and an increase in HDL levels compared to the diabetic control group.
Conclusion
ARA-290 peptide may induce advantageous action as an analog of erythropoietin. Researchers suggest the peptide exerts potential by activating the innate repair receptor (primary pathway) and inhibiting the TRPV1 cycle, possibly resulting in reduced pain sensations. Continued research and clinical trials are ongoing to explore the full potential of the peptide fully.
NOTE: These products are intended for laboratory research use only. This peptide is not intended for personal use. Please review and adhere to our Terms and Conditions before ordering.
References:
- Dahan, A., Dunne, A., Swartjes, M., Proto, P. L., Heij, L., Vogels, O., van Velzen, M., Sarton, E., Niesters, M., Tannemaat, M. R., Cerami, A., & Brines, M. (2013). ARA 290 improves symptoms in patients with sarcoidosis-associated small nerve fiber loss and increases corneal nerve fiber density. Molecular medicine (Cambridge, Mass.), 19(1), 334–345. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3883966/
- Image 1 credit: National Center for Biotechnology Information (2023). PubChem Compound Summary for CID 91810664, Cibinetide. Retrieved May 3, 2023, from https://pubchem.ncbi.nlm.nih.gov/compound/Cibinetide.
- Brines M, Cerami A. The receptor that tames the innate immune response. Mol Med. 2012 May 9;18(1):486-96. https://pubmed.ncbi.nlm.nih.gov/22183892
- Mashreghi, Moeen, et al. “An in Vivo Investigation on the Wound-Healing Activity of Specific Ligand for the Innate Repair Receptor, ARA290, Using a Diabetic Animal Mode.” Europe PMC, 2023. europepmc.org/article/ppr/ppr610510
- Winicki NM, Nanavati AP, Morrell CH, Moen JM, Axsom JE, Krawczyk M, Petrashevskaya NN, Beyman MG, Ramirez C, Alfaras I, Mitchell SJ, Juhaszova M, Riordon DR, Wang M, Zhang J, Cerami A, Brines M, Sollott SJ, de Cabo R, Lakatta EG. A small erythropoietin-derived non-hematopoietic peptide reduces cardiac inflammation, attenuates age-associated declines in heart function, and prolongs healthspan. Front Cardiovasc Med. 2023 Jan 18;9:1096887. doi: 10.3389/fcvm.2022.1096887. PMID: 36741836; PMCID: PMC9889362. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9889362/
- O’Leary OE, Canning P, Reid E, Bertelli PM, McKeown S, Brines M, Cerami A, Du X, Xu H, Chen M, Dutton L, Brazil DP, Medina RJ, Stitt AW. The vasoreparative potential of endothelial colony-forming cells in the ischemic retina is enhanced by cibinetide, a non-hematopoietic erythropoietin mimetic. Exp Eye Res. 2019 May;182:144-155. https://pubmed.ncbi.nlm.nih.gov/30876881/
- Yan L, Zhang H, Gao S, Zhu G, Zhu Q, Gu Y, Shao F. EPO Derivative ARA290 Attenuates Early Renal Allograft Injury in Rats by Targeting NF-κB Pathway. Transplant Proc. 2018 Jun;50(5):1575-1582. https://pubmed.ncbi.nlm.nih.gov/29880388/
- Nairz, M., Haschka, D., Dichtl, S. et al. Cibinetide dampens innate immune cell functions, thus ameliorating the course of experimental colitis. Sci Rep 7, 13012 (2017). https://doi.org/10.1038/s41598-017-13046-3
